Probiotics may benefit infants born by cesarean or exposed to antibiotics

Maternal bacterial colonization at birth
At birth, infants are colonized by maternal bacteria, which plays an important role for the developing immune system,1 and may have a long term impact on the infant’s health.2
Bifidobacteria and infant immunity
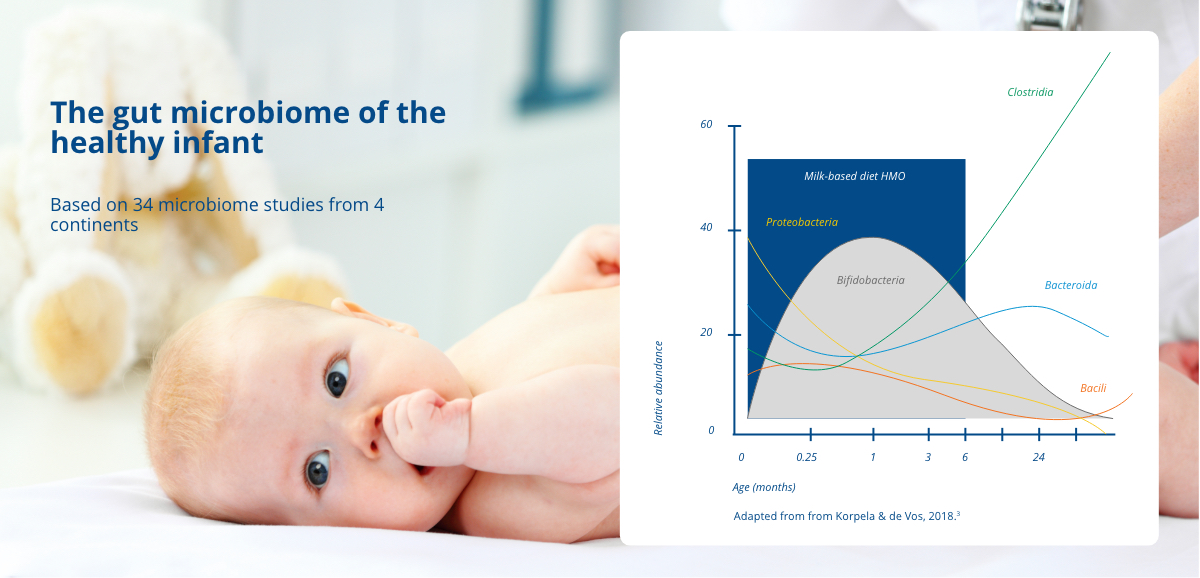
In the first few weeks of life, bifidobacteria is the most abundant group of bacteria in a healthy infant gut.3 Bifidobacteria have a significant role in the development and maturation of a healthy immune system, as they have been observed to interact with immune cells, modulating innate and adaptive immune processes.4 Through this process, a balanced and tolerogenic immune system evolves, reducing the risk of symptoms related to atopic or autoimmune diseases.2
If the composition of the infant intestinal microbiota is disturbed, there is an altered ratio of large bacteria taxa, and less bifidobacteria – this is called dysbiosis, and has been associated with an increased risk for some diseases later in life.1, 2 Dysbiosis occurs frequently in the initial colonization of the newborn’s intestine and has two common causes: 1) cesarean delivery, 2) use of antibiotics.
Cesarean delivery disruption to infant gut colonization
Infants born via cesarean delivery bypass the critical vertical transmission of maternal bacteria, especially bifidobacteria, that contribute to the formation of the infant gut microbiota.5 These infants have a much lower amount of bifidobacteria compared to vaginally delivered infants,2 and, thus, have a greater proportion of bacilli and enterobacteria.3 Lower numbers of bifidobacteria are associated with increased risk of obesity later in life, colic and atopic diseases.6
Antibiotic exposure disruption to infant gut colonization
Another common disturbance to infant microbiota is the use of antibiotics. Perinatal antibiotic exposure may compromise the microbiota's normal formational pattern.5 Whether pre- or post-natal, antibiotic exposure may decrease bifidobacteria and increase other species, resulting in a shift away from bifidobacteria’s important dominance.5 This disruption of the normal microbiota succession may impact infant development, and the use of antibiotics in early life has been associated with future allergic diseases, obesity, and the associated health risks.7

Probiotics contribute to optimal dominant prevalence of bifidobacteria in infants
In the perinatal period, bifidobacterial probiotic supplementation can contribute to bifidobacteria in the gut microbiota, supporting optimal dominant prevalence of bifidobacteria.6, 8 In a three-arm, randomized, double-blind, controlled study, new born infants either received Bifidobacterium, BB-12® (hereafter referred to by use of the trademark BB-12®) supplemented formula, received standard infant formula, or were breastfed. At 1 month of age, supplementation with BB-12® increased bifidobacteria colonization to a prevalence that was similar to that of breastfed infants, but significantly higher than the standard infant formula group.8 Further, in a 28-day clinical study of BB-12® probiotic supplementation in infants with colic, baseline vs. end-of-treatment microbiota analyses demonstrated that in those infants who responded to the BB-12® supplementation, there was a significant increase in the abundance of bifidobacteria. Additionally, improvement in symptoms of colic in the BB-12® treated group were significantly greater than in the control group.9 Click to read about probiotics and colic.
Other probiotic strains have also shown beneficial effects when used as probiotic supplementation in early infancy, such as supporting immune and gastrointestinal health.10, 11, 12, 13 For instance, supplementation with Lactobacillus rhamnosus, LGG® (hereafter referred to by use of the trademark LGG®) has been associated with shortened occurrence of acute diarrhea10, 13 and reduced incidence of gastrointestinal tract11 and respiratory tract infections.11, 12
Therefore, probiotic supplementation during early life is important as it may help balance the infant gut microbiota, which studies suggest can impact the incidence of colic,9 diarrhea,10, 13 respiratory tract infections,11, 12 and gastrointestinal tract infections11 as well as obesity and atopic disease later in life.6
Click to read about LGG®, BB-12®, probiotics and infant colic, or probiotic supplementation during pregnancy and breastfeeding.
BB-12® and LGG® are registered trademarks of Chr. Hansen A/S.
The article is provided for informational purposes regarding probiotics and is not meant to suggest that any substance referenced in the article is intended to diagnose, cure, mitigate, treat, or prevent any disease.
References Open Close
1. Gensollen T, et al. How colonization by microbiota in early life shapes the immune system. Science. 2016;352(6285):539-44. (PubMed)
2. Walker WA. The importance of appropriate initial bacterial colonization of the intestine in newborn, child, and adult health. Pediatr Res. 2017;82(3):387-95. (PubMed)
3. Korpela K, de Vos WM. Early life colonization of the human gut: microbes matter everywhere. Curr Opin Microbiol. 2018;44:70-8. (PubMed)
4. Ruiz L, et al. Bifidobacteria and Their Molecular Communication with the Immune System. Front Microbiol. 2017;8:2345-. (PubMed)
5. Korpela K, et al. Childhood BMI in relation to microbiota in infancy and lifetime antibiotic use. Microbiome. 2017;5(1):26. (PubMed)
6. Rautava S. Microbial Composition of the Initial Colonization of Newborns. In: Isolauri E, et al., editors. Intestinal Microbiome: Functional Aspects in Health and Disease 2017. (PubMed)
7. Korpela K, et al. Association of Early-Life Antibiotic Use and Protective Effects of Breastfeeding: Role of the Intestinal Microbiota. JAMA Pediatr. 2016;170(8):750-7. (PubMed)
8. Langhendries JP, et al. Effect of a fermented infant formula containing viable bifidobacteria on the fecal flora composition and pH of healthy full-term infants. J Pediatr Gastroenterol Nutr. 1995;21(2):177-81. (PubMed)
9. Nocerino R, et al. The therapeutic efficacy of Bifidobacterium animalis subsp. lactis BB-12® in infant colic: A randomised, double blind, placebo-controlled trial. Aliment Pharmacol Ther. 2019. (PubMed)
10. Isolauri E, et al. A human Lactobacillus strain (Lactobacillus casei sp strain GG) promotes recovery from acute diarrhea in children. Pediatrics. 1991;88(1):90-7. (PubMed)
11. Hojsak I, et al. Lactobacillus GG in the prevention of nosocomial gastrointestinal and respiratory tract infections. Pediatrics. 2010;125(5):e1171-7. (PubMed)
12. Hojsak I, et al. Lactobacillus GG in the prevention of gastrointestinal and respiratory tract infections in children who attend day care centers: a randomized, double-blind, placebo-controlled trial. Clin Nutr. 2010;29(3):312-6. (PubMed)
13. Aggarwal S, et al. Lactobacillus GG for treatment of acute childhood diarrhoea: an open labelled, randomized controlled trial. Indian J Med Res. 2014;139(3):379-85. (PubMed)
What are
probiotics?
Learn more about what probiotics are and how they can benefit your health
Clinical
studies
Learn how to review the evidence when considering probiotic strains and effects
Our
strains
Read more about some of the world’s most documented probiotic strains and their diverse, beneficial health effects



